 The EUA means that the Innova lateral flow test can be used by a member of the public, with no previous experience of testing, in their own home or another community setting such as a place of work. 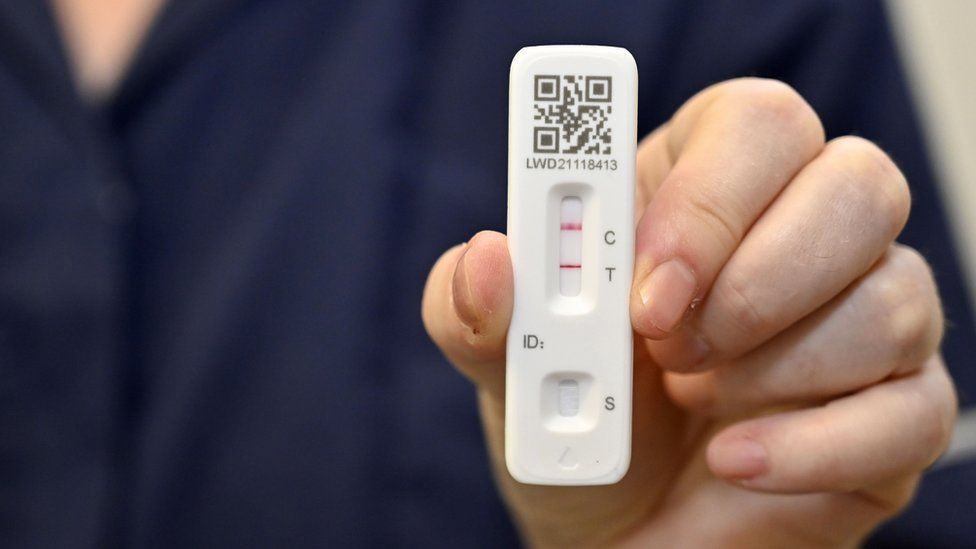
In December 2020, the Medicines and Healthcare products Regulatory Agency (MHRA) issued an exceptional use authorisation (EUA) to the Department of Health and Social Care to allow the use of the Innova lateral flow self-test to detect infection in asymptomatic individuals who otherwise would not be tested.Īn EUA is when a manufacturer applies to supply a medical device that does not comply with the law to protect a patient’s health if there is no legitimate alternative available. The benefit of lateral flow tests is that most tests generate easy-to-understand results in under half an hour and can be used at the point of care rather than sent to a lab to process. Lateral flow antigen tests are tests that can process COVID-19 samples quickly without the need for laboratory equipment. They differ from polymerase chain reaction (PCR) tests, which look for genetic material from the virus and are generally more sensitive. Sign in now if you’re an RPS member or subscriber.īecome a member of the RPS or subscribe to gain unlimited access. Journal of Pharmaceutical Health Services Research. 
International Journal of Pharmacy Practice.Antimicrobial resistance and stewardship. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
